| Research |
| Location:Home > Research > Research Progress |
| New Advances in Sensitive Polymer Membrane Based Potentiometric Sensors |
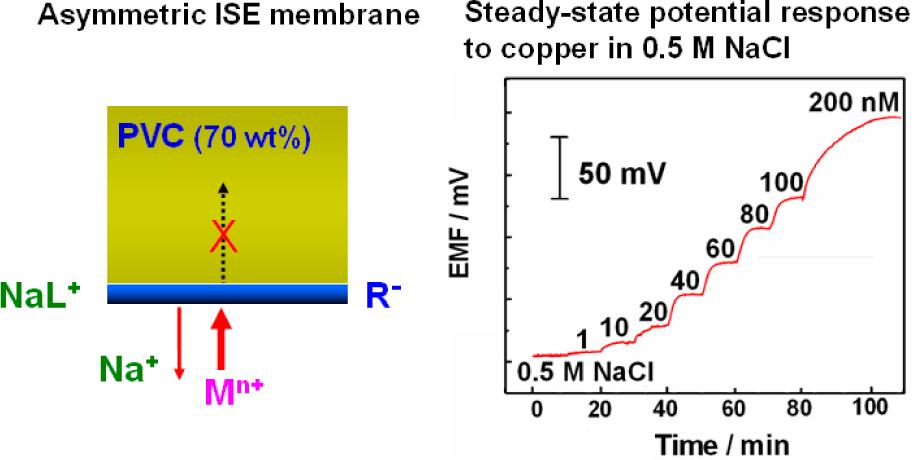
The first polymeric membrane ion-selective electrode (ISE) was developed in 1960s. Large improvements have been made on its performance in the past decades. And now they have been widely used in clinical diagnosis and environmental analysis owing to their attractive features including excellent selectivity, ease of use, low cost, and high reliability. Systematic studies have been done by Qin Wei’s group (Key Laboratory of Coastal Zone Environmental Processes and Ecological Remediation, Chinese Academy of Sciences) in order to further improve the responsive characteristics of the ISE and expand the application areas of the potentiometric transducers: 1 Trace-level potentiometric detection in the presence of a high electrolyte background For a long time, applications of such ISEs are often limited to measurements with low levels of electrolyte background owing to the presence of interferences ions. Our group develops an asymmetric membrane rotating ISE configuration for trace-level potentiometric detection with a high-interfering background. By using the asymmetric membrane with an ion exchanger loaded on the membrane surface, the diffusion of the primary ions from the organic boundary layer into the bulk of the membrane can be effectively blocked; on the other hand, rotation of the membrane electrode dramatically reduces the diffusion layer thickness of the aqueous phase and significantly promotes the mass transfer of the primary ions to the sample membrane interface. The induced accumulation of the primary ions in the membrane boundary layer largely enhances the nonequilibrium potential response. By using copper as a model, the new concept offers a subnanomolar detection limit for potentiometric measurements of heavy metals with a high electrolyte background of 0.5 M NaCl. New thoughts have been provided by this work for further development of trave-level heavy metal detection technologies. The corresponding research results have been published on Analytical Chemistry (Anal. Chem., 2012, 84 (24), 10509–10513;IF=5.856).
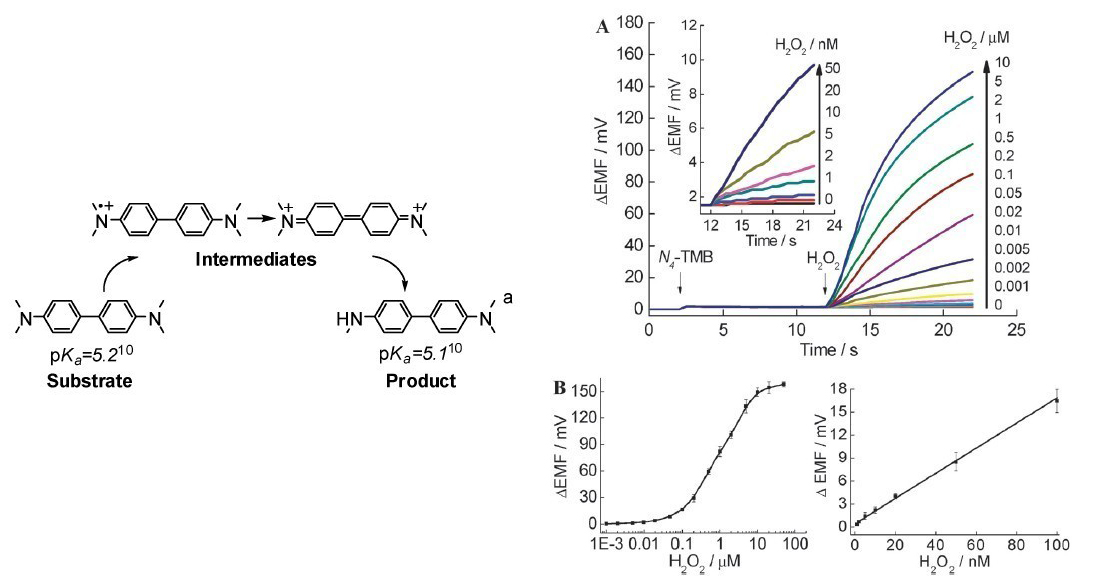
2 Reactive intermediates-induced potential responses of a polymeric membrane electrode for ultrasensitive potentiometric biosensing While the potential responses of stable reactants or productsare usually used by traditional potentiometric biosensing for detection, for a long time, a number of analytically important reactions which can’t give out ionic reactants or products are considered as useless in potentiometic detection. It was found by our group that reactive intermediates rather than stable reactants or products could induce large potential responses on an appropriately formulated polymeric membrane electrode, so the corresponding reactions could be detected. By using horseradish peroxidase-H2O2-N,N',N,N'-tetramethylbenzidine as a model, DNNS doped polymer membrane can be used for ultrasensitive potentiometric biosensing. The detection limit toward H2O2 obtained by the intermediate-sensitive electrode is 10-9M, which is three orders of magnitude lower than those obtained by traditional ISEs for stable ions. This novel methodology can satisfy the requirements for H2O2 detection in environmental water samples including seawater and rainwater. The corresponding research results have been published on Chemical Communication (Chem. Commun. 2012, 48, 4073-4075;IF= 6.169).
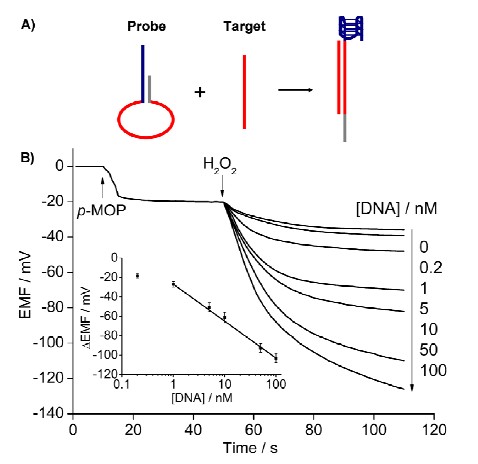
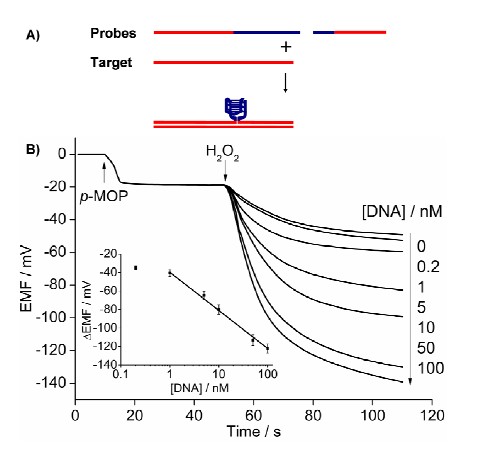
3 Novel biosensing based on the potentiometric response of neutral phenols The response of phenols on the polymeric membrane electrodes is an important discovery in the area of potentiometric analytical chemistry in the last 20 years. Since the low sensitivity, this response has never been used in environmental or biological analysis. Howerer, it was found my our group that electrically neutral oligomeric phenols could induce highly sensitive potential responses on quaternary ammonium salt-doped polymeric membrane electrodes owing to their high lipophilicities. In the presence of G-quadruplex/hemin DNAzyme and H2O2, monomeric phenols can be condensed into oligomeric phenols, and hence the first potentiometric transducer for G-quadruplex/hemin DNAzyme-based biosensing has been developed based on the principles proposed above. The proposed polymeric membrane-based potentiometric sensor promises to be a competitive transducer relative to its colorimetric and fluorometric counterparts owing to its features such as high sensitivity, low cost, and resistance to color and turbid interferences. This sensing platform can be applied in homogenous DNA hybridization detection and DNA damage analysis, and promises to be an effective transducer for the identification of genotoxicity of environmental pollutants. The corresponding research results will be published on Analytical Chemistry (Anal. Chem., 2013, DOI: 10.1021/ac3035629; IF=5.856).
|
| Appendix Download |
|
|