The mussel Mytilus galloprovincialis, a typical marine organism in intertidal zone, is widely distributed in many sea areas of China and the other parts of the world. Because of its high resistance to environmental pollutants (heavy metals, pathogenic microorganism etc.), M. galloprovincialis is usually used as a bioindicator to monitor the change of marine environment. At present, the adaptive mechanism of the mussel to the environmental factors of intertidal zone has received much concern.
The environmental toxicology research team has characterized two goose-type lysozyme genes from M. galloprovincialis. They have showed that the recombinant goose-type lysozymes could kill several pathogens which were usually found in sea waters. In addition, goose-type lysozymes have been proved to participate in immune response against pathogens invasion. By protein function and bioinformatics analysis, the researchers found that the physiological functions of the two goose-type lysozymes have diversified under positive selection during the evolution. All the above findings will bring lights to understand the mechanism of mussels adapting to intertidal environment stress.
This work titled
"Two goose-type lysozymes in Mytilus galloprovincialis: possible function diversification and adaptive evolution" has been published in
PLoS One, and Dr. WANG Qing is the first author of this article.
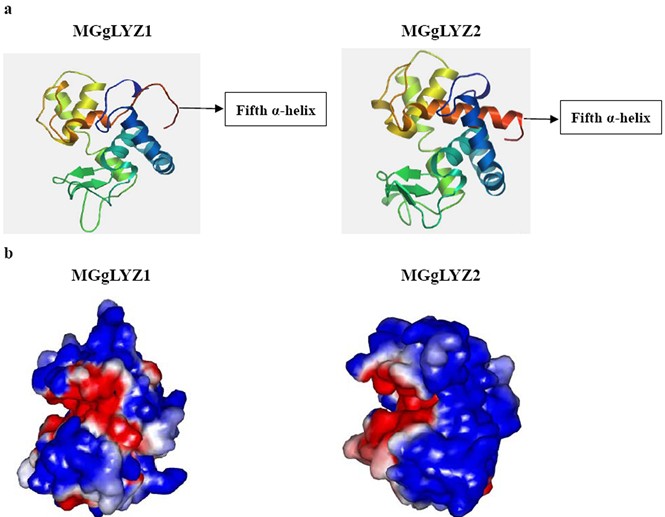
Figure1:Predicted three-dimensional structure of MGgLYZ1 and MGgLYZ2.
a: Three-dimensional structure predicted by SWISSMODEL. b: Electrostatic surface potentials of MGgLYZ1 and MGgLYZ2. The color runs from intense red (lowest) to intense blue (highest) potential.(Image by WANG Qing et al)
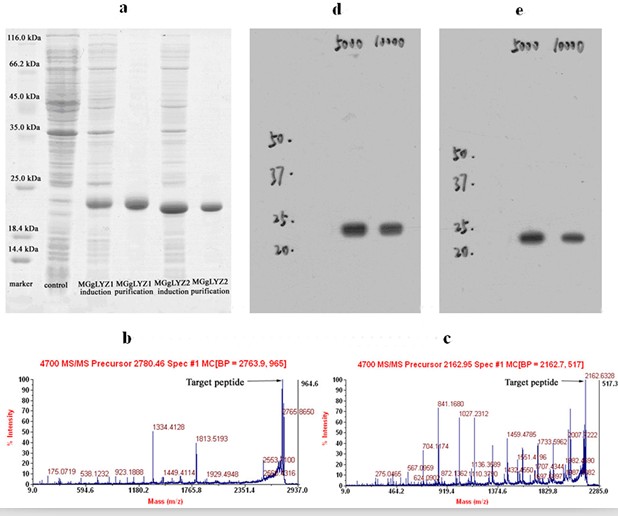
Figure 2: Analysis of recombinant MGgLYZ1 and MGgLYZ2 proteins and antibodies. (a) SDS-PAGE analysis, LC-ESI-MS/MS identification of MGgLYZ1 (b) and MGgLYZ2 (c), western blotting analysis of MGgLYZ1 (d) and MGgLYZ2 (e) polyclonal antibodies. (Image by WANG Qing et al)
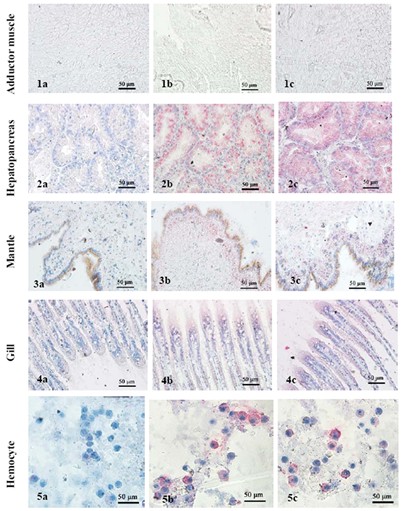
Figure 3: Immunolocalization of MGgLYZ1 and MGgLYZ2 in different tissues. Negative control of muscle (1a), hemocytes (2a), mantle (3a), hepatopancreas (4a) and gill (5a). Localization of MGgLYZ1 in muscle (1b), hemocytes (2b), mantle (3b), hepatopancreas (4b) and gill (5b). Localization of MGgLYZ2 in muscle (1c), hemocytes (2c), mantle (3c), hepatopancreas (4c) and gill (5c). Positive signal was stained red and the other signals were stained blue. (Image by WANG Qing et al)